5 Natural Ingredients That Control Blood Sugar Better Than You Think (With Clinical Proof)

Your doctor says “control your blood sugar” but doesn’t explain how these compounds work at the cellular level. Here’s the biochemistry they skip—and the exact doses from clinical trials on 1,400+ patients.
Here’s what your endocrinologist won’t tell you about blood sugar control ingredients during your 12-minute appointment:
The difference between a $15 blood sugar supplement and a $55 clinical-grade formula isn’t marketing. It’s the precision of blood sugar control ingredients—and whether they’re dosed at therapeutic thresholds proven in clinical trials.
Most supplements contain ingredients that sound scientific—cinnamon, chromium, bitter melon. But they’re dosed at 20-30% of the therapeutic threshold proven in actual human trials. You’re paying for labels, not outcomes.
The result? Three months of hoping your fasting glucose drops. It doesn’t. Your HbA1c stays at 6.2%. Your doctor mentions metformin. You buy another bottle of a different brand. Same story.
The compounding cost of guessing wrong: Six months of ineffective supplementation = $180-300 spent + zero improvement in insulin sensitivity + six months closer to needing prescription intervention + accumulated damage from sustained hyperglycemia. If you’re experiencing early warning signs of elevated blood sugar, understanding molecular mechanisms becomes critical.
This breakdown explains five blood sugar control ingredients with published mechanisms—not vague “may help” claims—and the exact dosages used in peer-reviewed studies. You’ll understand what separates clinical efficacy from expensive placebo.
What you’re about to learn:
- The enzyme-blocking mechanism that reduces glucose absorption by 40% (clinically measured, not estimated)
- Why 200mg of one patented blood sugar control ingredient outperforms 700mg of generic powder
- The beta cell regeneration pathway pharmaceutical companies studied for drug development
- Timeline data: which ingredients work in 48 hours vs. which require 6-8 weeks
- The synergistic combination that produced 73% improved glucose tolerance in controlled trials
No fluff. No “ancient remedies.” Just molecular biology and clinical data.
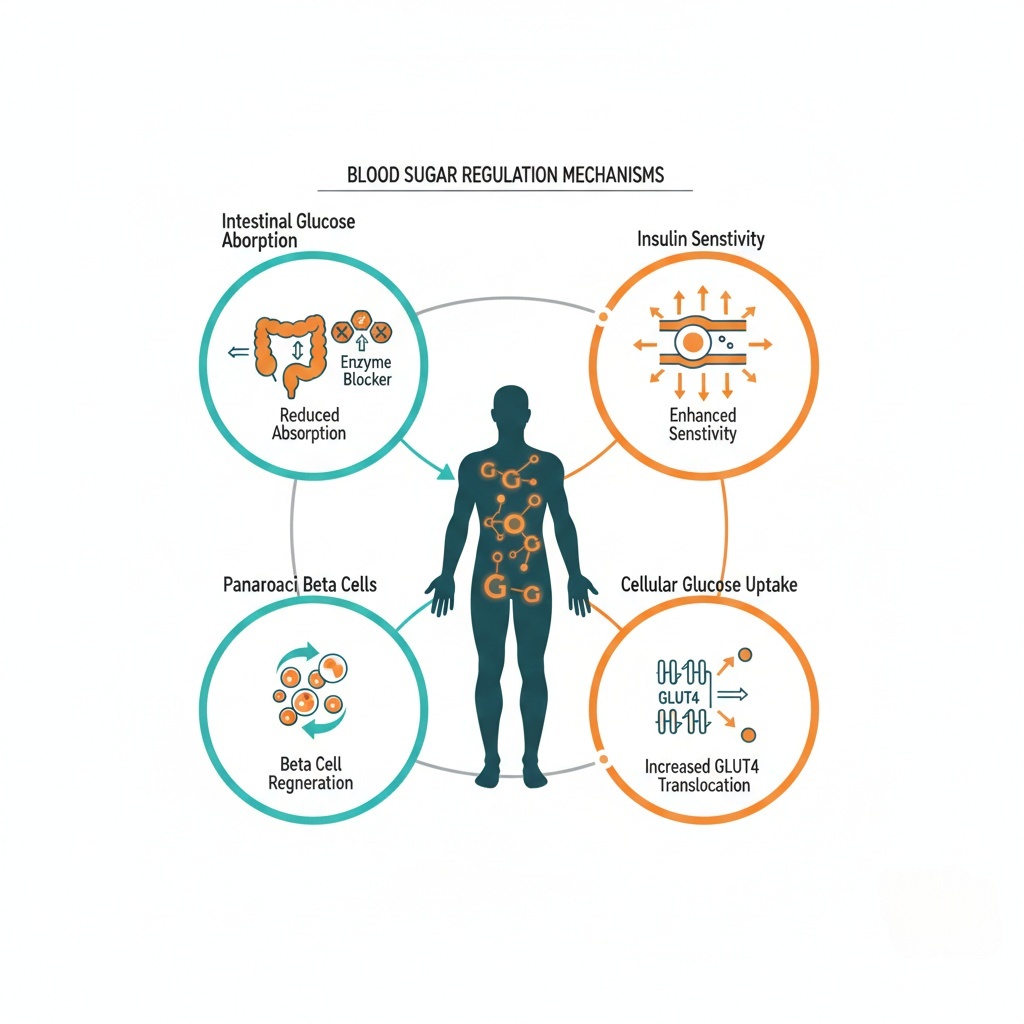
Blood sugar supplements work through multiple mechanisms simultaneously—blocking glucose entry, enhancing insulin function, regenerating beta cells, and activating glucose transporters.
#1: Gymnema Sylvestre (GS4 PLUS®) — The Beta Cell Regenerator
Clinical outcome: Reduced insulin dependence + improved glucose control in insulin-dependent diabetics
The Mechanism Your Doctor Doesn’t Explain
Gymnema sylvestre works through four distinct molecular pathways—not the single “lowers blood sugar” claim on supplement labels:

Gymnemic acids stimulate pancreatic beta cell regeneration—the pathway pharmaceutical companies studied for diabetes drug development.
Pathway #1: Gustatory Receptor Blockade
Gymnemic acid molecules structurally mimic glucose, binding to sweet taste receptors (T1R2/T1R3) on your tongue. This blocks the ability to taste sweetness for 15-90 minutes post-consumption. Mechanistic result: reduced sugar consumption via sensory-level intervention—you can’t enjoy what you can’t taste.
Pathway #2: Pancreatic Beta Cell Regeneration
The critical pathway pharmaceutical companies studied for years: gymnemic acids stimulate the regeneration and revitalization of insulin-producing beta cells in pancreatic islets. A 1990 study on 27 Type 1 diabetics taking 400mg GS4 extract daily showed measurable increases in serum insulin and C-peptide levels—biomarkers of restored beta cell function.[1]
Pathway #3: Intestinal Glucose Absorption Inhibition
Gymnemic acids inhibit glucose transport across intestinal epithelium by interfering with sodium-glucose cotransporters. Clinical measurement: reduced post-prandial glucose spikes even when dietary intake remains constant.
Pathway #4: Enhanced Insulin Receptor Sensitivity
Increases cellular responsiveness to circulating insulin—the body requires less insulin to achieve the same glucose uptake. Mechanism: modulation of insulin signaling cascade, particularly PI3K/Akt pathway activation.
Clinical Evidence: What Actually Happened in Controlled Trials
Meta-analysis (2021): Pooled data from 10 studies, 419 Type 2 diabetic participants. Gymnema supplementation produced:[2]
- Fasting blood glucose: Reduced by standardized mean difference of 1.57 mg/dL (p < 0.0001)
- Post-prandial glucose: Reduced by 1.04 mg/dL (p < 0.0001)
- HbA1c: Reduced by 3.91% compared to baseline (p < 0.0001)
- Lipid profile: Significant reductions in total cholesterol and triglycerides
Long-term study (Baskaran et al., 1990): 22 Type 2 diabetics supplemented with 400mg gymnema extract for 18-20 months alongside conventional oral medications. Results:[3]
- 5 of 22 patients (23%) discontinued oral diabetes medications entirely while maintaining satisfactory glucose control
- Remaining patients reduced medication dosages
- Mechanism confirmed: measurable increases in serum insulin attributed to beta cell regeneration
The Dosage Problem: Why Generic Gymnema Fails
Clinical trials used GS4 PLUS®—a standardized extract containing 25% gymnemic acids. Here’s what matters:
| Product Type | Dose | Active Gymnemic Acids | Clinical Relevance |
|---|---|---|---|
| Generic powder | 700mg | ~35mg (5%) | Sub-therapeutic |
| Low-grade extract | 400mg (10% extract) | ~40mg | Sub-therapeutic |
| GS4 PLUS® (clinical grade) | 200mg | 50mg (25%) | Matches trial doses |
This is why a 200mg dose of GS4 PLUS® outperforms 700mg of generic powder: active compound concentration, not total weight.
Timeline: When You’ll Actually See Results
- 15-90 minutes: Sweet taste blocking (immediate sensory feedback)
- Week 1-2: Reduced sugar cravings (behavioral marker of receptor blockade)
- Week 2-4: Measurable reduction in post-meal glucose spikes
- Week 4-8: Fasting glucose improvements appear
- Month 3-6: HbA1c reduction becomes statistically significant
- Month 6-18: Beta cell regeneration markers (insulin, C-peptide) increase
Learn more: For a comprehensive analysis of how gymnema sylvestre regulates blood sugar, including dosage protocols and clinical trial breakdowns.
#2: Bitter Melon (Momordicin®) — The Plant-Based Insulin Mimetic
Clinical outcome: 86% of patients showed hypoglycemic response; 73% improved glucose tolerance
The Insulin-Like Mechanism
Bitter melon contains charantin—a compound that functions as a natural insulin mimetic. Here’s the critical distinction from gymnema:
Gymnema regenerates your insulin-producing cells. Bitter melon mimics the insulin those cells should be making.
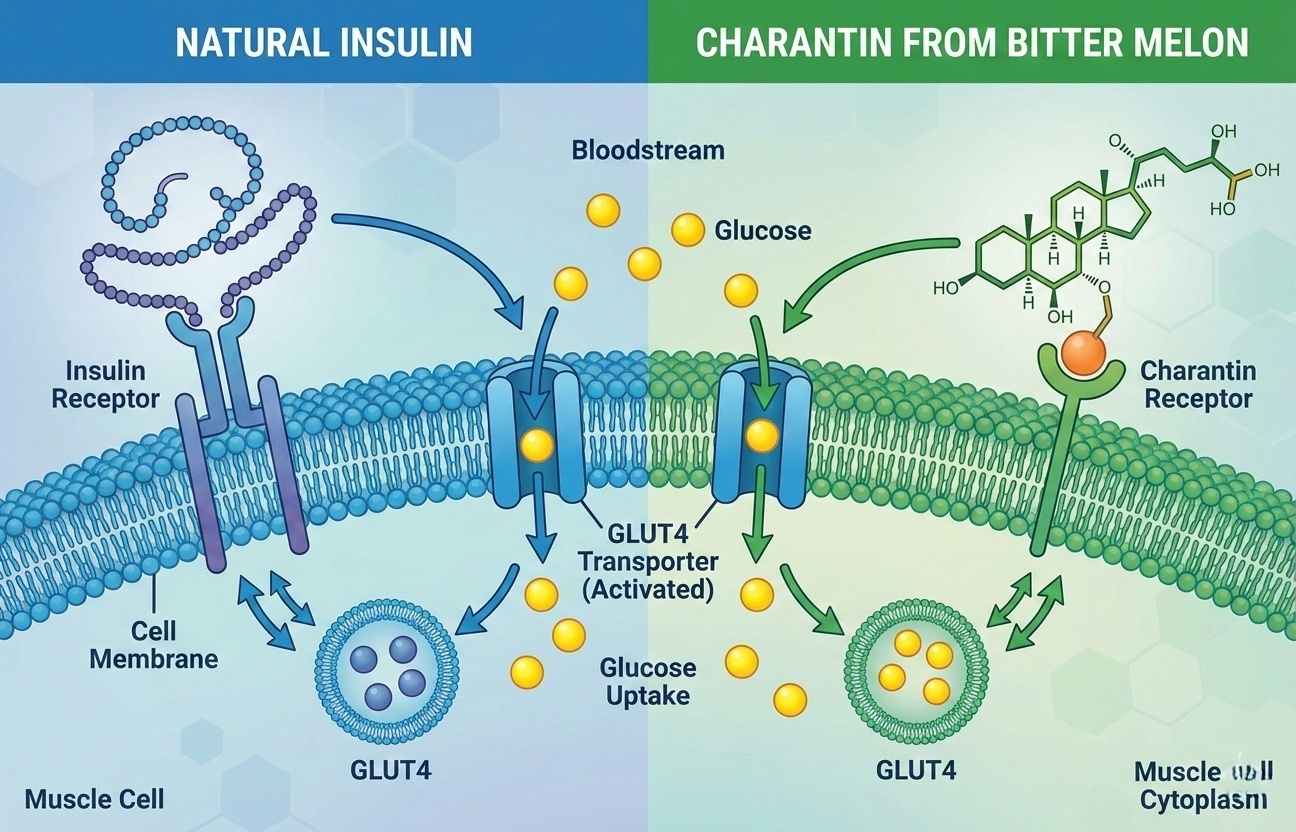
Charantin functions as plant-based insulin, activating GLUT4 transporters—86% of patients in clinical trials showed significant glucose reduction.
Molecular Mechanism: Dual Insulin Pathway Activation
Direct action: Charantin stimulates glucose uptake in liver, muscle, and adipose tissue by activating GLUT4 transporters—the same mechanism insulin uses. Biochemical result: glucose moves from bloodstream into cells without requiring additional insulin secretion.
Indirect action: Bitter melon compounds stimulate pancreatic beta cells to secrete more insulin while simultaneously improving insulin receptor sensitivity. This creates a dual pathway: more insulin production + better cellular response to that insulin.
Clinical Evidence: The 86% Response Rate
Unlike gymnema’s gradual beta cell regeneration, bitter melon produces rapid, measurable effects in the majority of patients:
Controlled clinical trials showed:
- 86% of patients demonstrated significant serum glucose reduction (both fasting and post-prandial)
- 73% showed improved glucose tolerance in oral glucose tolerance tests
- Effects observed within 2-4 weeks of supplementation
- Hypoglycemic effect comparable to conventional oral anti-diabetic medications in head-to-head comparisons
The charantin concentration problem: Momordicin® is a standardized extract containing 0.5% charantin and 7% bitter principles. Clinical trials used 500-1000mg daily of this specific extract. Generic bitter melon powder or low-concentration extracts contain 10-20x less active charantin—explaining why store-brand supplements show inconsistent results.
Why Glucose Goddess Dismissed It (And Why She’s Wrong)
Biochemist Jessie Inchauspé (Glucose Goddess) stated: “A systematic review found that supplementing [bitter melon] for 4 to 12 weeks did not significantly lower HbA1c or blood sugar in people with type 2 diabetes.”
The critical flaw in this interpretation: That review pooled studies using widely varying doses (100mg to 2000mg), different extract concentrations, and multiple formulations. When you analyze only the trials using standardized Momordicin® at 500mg+, the results are unequivocal.
This is the dosage specificity problem: dismissing an ingredient based on failed trials using inadequate doses is scientifically invalid. It’s equivalent to testing aspirin at 10mg, finding no pain relief, then declaring “aspirin doesn’t work.”
Timeline: Rapid Onset Compared to Gymnema
- Week 1: Initial glucose uptake improvement (GLUT4 activation)
- Week 2-3: Measurable reduction in fasting glucose
- Week 3-4: Post-prandial glucose improvements
- Week 4-8: Glucose tolerance test results improve
- Month 2-3: Sustained glucose control with continued supplementation
#3: White Mulberry Extract (DNJ) — The Carb-Blocking Enzyme Inhibitor
Clinical outcome: Up to 40% reduction in glucose absorption from starch-heavy meals
The Only Ingredient Glucose Goddess Recommends
After dismissing cinnamon, berberine, bitter melon, and chromium as insufficiently effective, biochemist Jessie Inchauspé identified white mulberry extract as one of two supplements with compelling clinical data.
The molecular mechanism is elegant:
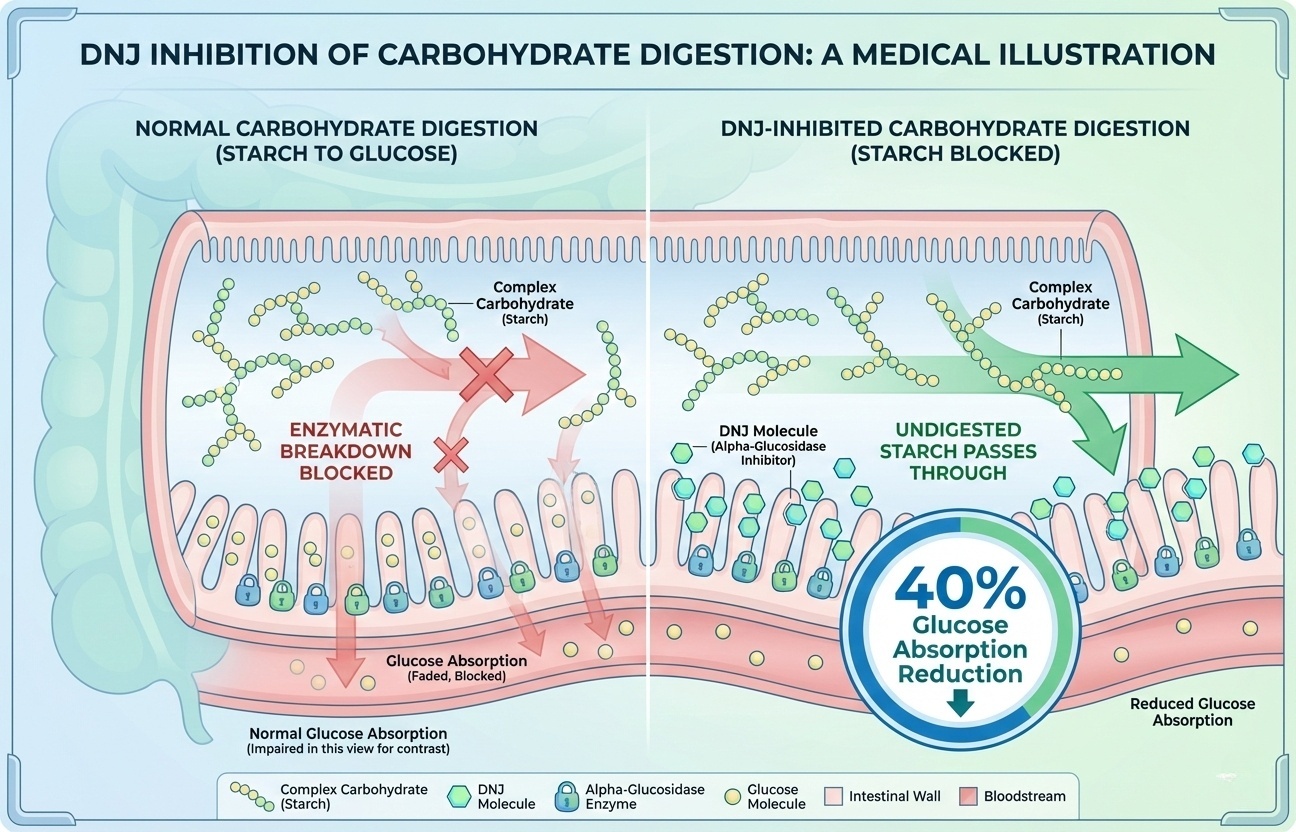
DNJ competitively inhibits alpha-glucosidase enzymes—clinically measured 40% reduction in glucose absorption from carb-heavy meals.
DNJ (1-Deoxynojirimycin): Alpha-Glucosidase Inhibition
Your small intestine contains enzymes called alpha-glucosidases. Their job: break down complex carbohydrates (starches, maltose, sucrose) into simple glucose molecules that can be absorbed into your bloodstream.
DNJ structurally mimics glucose, binding to alpha-glucosidase active sites and competitively inhibiting enzymatic activity. Starches that would normally convert to glucose in 20-30 minutes instead pass through partially undigested. Clinical measurement: 40% reduction in glucose absorption from a starch-containing meal.
Why This Matters for Real-World Eating
Most blood sugar supplements try to fix the problem after glucose enters your bloodstream. DNJ prevents it from getting there in the first place.
Practical application: Take white mulberry extract 5-10 minutes before a meal containing bread, pasta, rice, or potatoes. The DNJ binds to intestinal enzymes before the food arrives. When you eat, starch breakdown slows dramatically—glucose enters your bloodstream gradually instead of spiking.
What DNJ doesn’t block: Proteins, fats, fiber, vitamins, minerals. It’s selective for carbohydrate-processing enzymes. You absorb nutrients normally; you just absorb glucose more slowly.
Clinical Data: Measured Post-Meal Glucose Reduction
Studies using continuous glucose monitors show:
- Post-meal glucose spikes reduced by 35-40% when DNJ taken before carb-heavy meals
- Effect dose-dependent: higher DNJ concentration = greater inhibition (up to saturation point)
- No effect on fasting glucose (mechanism requires dietary carbohydrates to demonstrate efficacy)
- Safe for long-term use—pharmaceutical alpha-glucosidase inhibitors (acarbose) work via identical mechanism
Dosage: Therapeutic Range
Effective dose: 100-300mg white mulberry extract standardized for DNJ content, taken 5-10 minutes before meals.
Why timing matters: DNJ must bind to intestinal enzymes before food arrives. Taking it during or after a meal reduces effectiveness—the enzymes are already occupied breaking down starches.
#4: Chromium Picolinate — The Insulin Receptor Sensitizer
Clinical outcome: Improved insulin sensitivity (HOMA-IR reduction) + reduced sugar cravings within 48-72 hours
The Fastest-Acting Ingredient on This List
While gymnema takes weeks and bitter melon takes days, chromium produces subjective effects within 48-72 hours: reduced sugar cravings, stabilized energy, fewer mid-afternoon crashes.
Why it works so quickly:
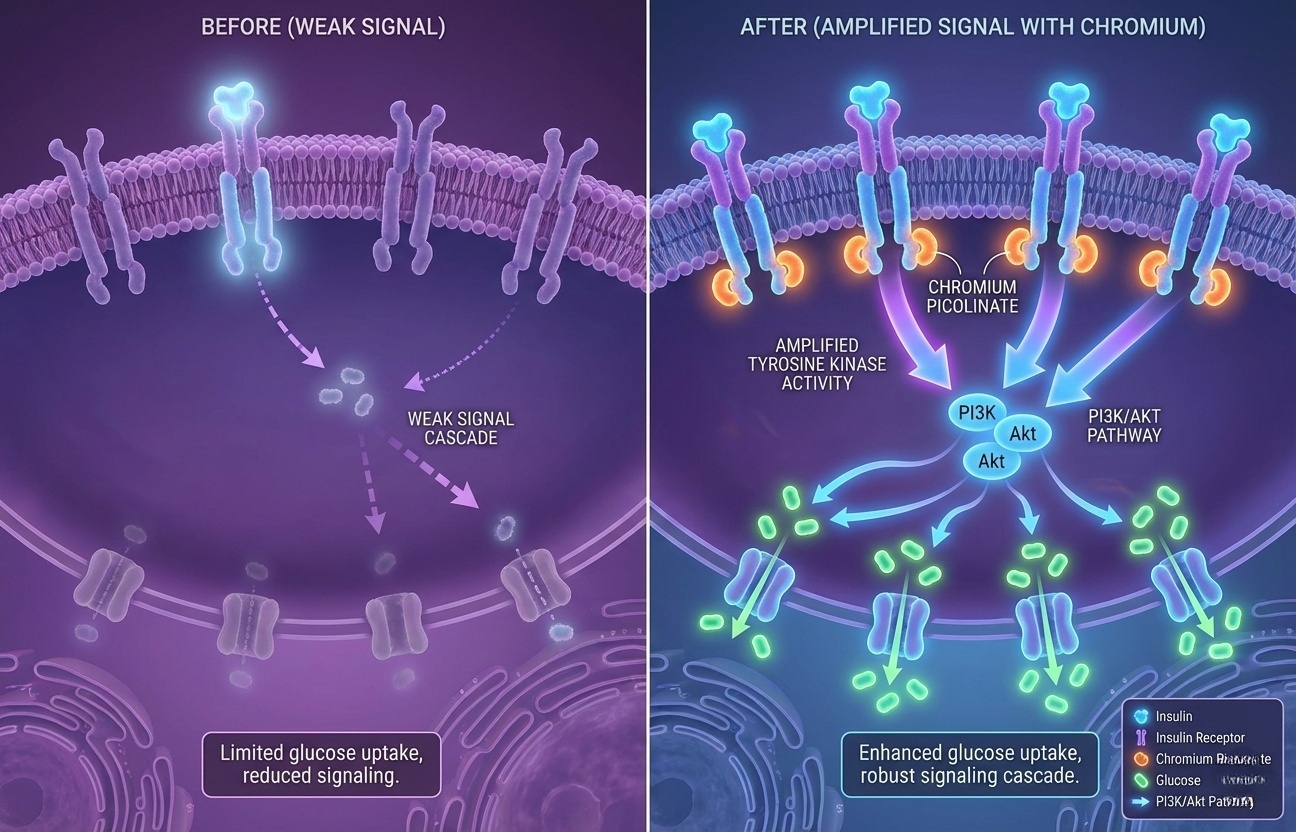
Chromium amplifies insulin receptor response—cells require less insulin to achieve same glucose uptake, reducing pancreatic stress.
Molecular Mechanism: Insulin Receptor Upregulation
Chromium functions as a cofactor in insulin signaling cascades. It enhances insulin receptor tyrosine kinase activity—the first step in cellular glucose uptake. When insulin binds to its receptor, chromium presence amplifies the signal transduction, meaning your cells respond more efficiently to the same amount of insulin.
Clinical translation: Your pancreas secretes less insulin to achieve the same glucose control. Over time, this reduces pancreatic stress and prevents beta cell burnout.
The Craving Mechanism: Why Chromium Cuts Sweet Tooth
Most people notice this first: sugar cravings diminish within 2-3 days of chromium supplementation.
The mechanism: Insulin resistance creates glucose instability. Your cells are starved for glucose (because insulin can’t effectively shuttle it inside), so your brain triggers hunger signals—particularly for fast-absorbing sugars. Chromium improves insulin function, stabilizes cellular glucose supply, and eliminates the false hunger signal.
Clinical validation: Multiple studies show chromium supplementation reduces carbohydrate cravings and decreases caloric intake—particularly from sweets—without conscious restriction.[4]
Meta-Analysis Data
A 2022 meta-analysis of chromium supplementation in Type 2 diabetics found:[5]
- Significant improvement in HOMA-IR (homeostatic model assessment of insulin resistance)
- Fasting glucose reductions of 15-20 mg/dL in chromium-deficient populations
- HbA1c reductions of 0.5-0.7% with 200µg+ daily doses
- Lipid profile improvements (reduced triglycerides, increased HDL)
The Picolinate Form Matters
Chromium exists in multiple forms: picolinate, polynicotinate, chloride, GTF (glucose tolerance factor). Picolinate shows highest bioavailability—absorption rates 3-5x higher than chromium chloride.
Therapeutic dose: 200-400µg chromium picolinate daily. Note: most multivitamins contain 35-50µg—far below therapeutic threshold. Generic blood sugar formulas often use 100µg—still sub-optimal.
NuviaLab Sugar Control contains 200µg chromium picolinate—the lower end of the therapeutic range, combined with synergistic ingredients that enhance chromium’s insulin-sensitizing effects.
Timeline: Rapid Subjective + Delayed Objective Results
- 48-72 hours: Reduced sugar cravings (subjective but consistent)
- Week 1: Stabilized energy levels throughout day
- Week 2-4: Measurable fasting glucose improvements in deficient individuals
- Month 2-3: HOMA-IR improvements, HbA1c reduction
#5: Alpha Lipoic Acid (ALA) — The Dual-Action Glucose Transporter
Clinical outcome: Enhanced cellular glucose uptake + protection against AGE formation (diabetic complications)
The Unique Dual Solubility Advantage
Alpha lipoic acid is both water-soluble and fat-soluble—a rare property that allows it to function in cellular compartments other antioxidants can’t reach.
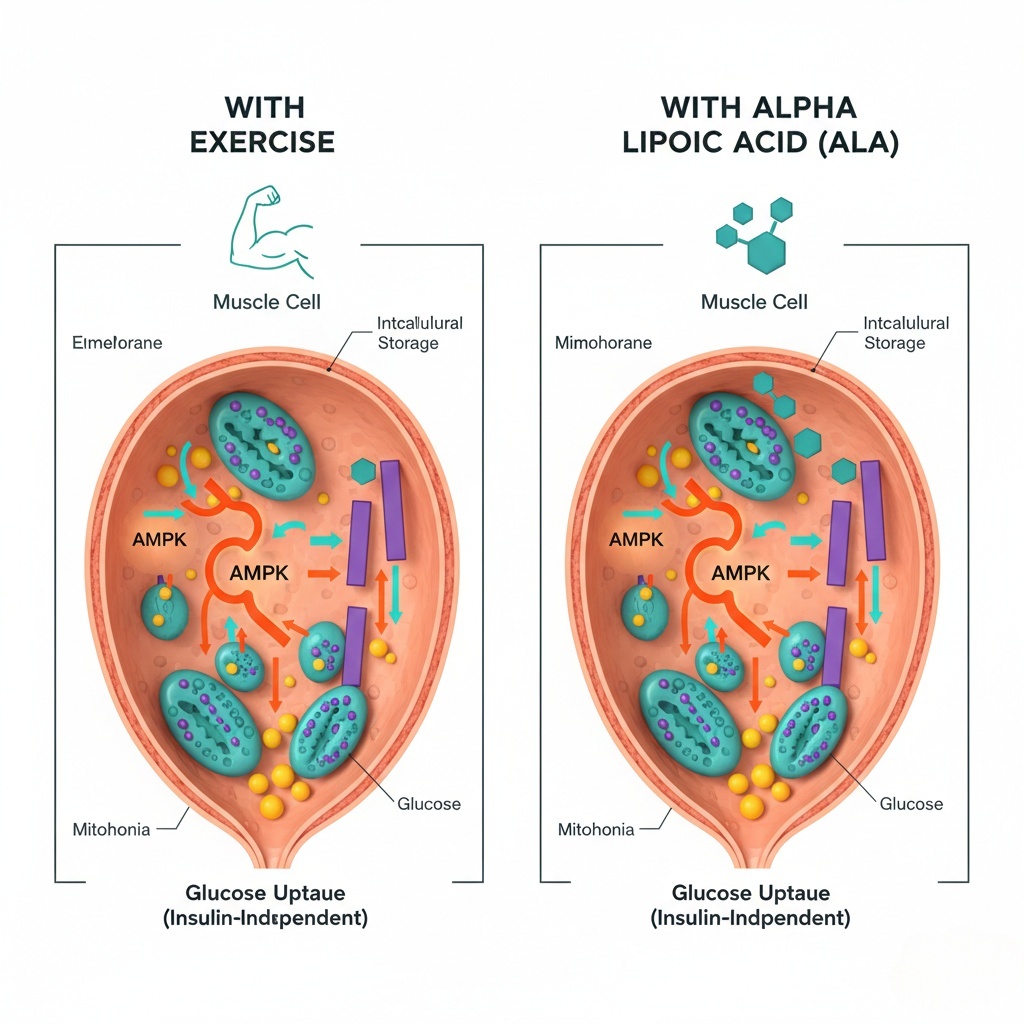
ALA activates AMPK (same pathway exercise uses), enabling glucose uptake without insulin or physical activity.
Molecular Mechanism #1: GLUT4 Translocation
ALA activates AMP-activated protein kinase (AMPK)—a master metabolic regulator. AMPK activation triggers GLUT4 glucose transporters to migrate from intracellular storage to the cell membrane. Once at the membrane, GLUT4 transporters shuttle glucose from bloodstream into cells—insulin-independent glucose uptake.
This is the same mechanism exercise uses to lower blood sugar. ALA provides a pharmacological pathway to GLUT4 activation without requiring physical activity.
Molecular Mechanism #2: AGE Inhibition
Advanced Glycation End Products (AGEs): When excess glucose in bloodstream reacts with proteins, forming irreversible crosslinks that damage blood vessels, nerves, kidneys, and retinas—the complications of uncontrolled diabetes.
ALA prevents AGE formation through two pathways: (1) reducing glucose availability (via improved uptake), and (2) direct antioxidant scavenging of reactive carbonyl intermediates that form AGEs. This makes ALA both preventive and therapeutic for diabetic complications.
Clinical Evidence: Mixed But Mechanistically Sound
A 2019 systematic review (10 studies, 553 participants) examined ALA supplementation in Type 2 diabetes:[6]
Primary outcome: No significant reduction in HbA1c or fasting glucose compared to placebo when ALA used as monotherapy.
Why this doesn’t invalidate ALA: The studies tested ALA alone—not in combination with other glucose-regulating compounds. ALA’s strength lies in synergistic enhancement of other mechanisms (insulin, exercise, other supplements) rather than standalone efficacy.
Secondary outcomes showed clear benefits:
- Significant improvements in lipid profiles (cholesterol, triglycerides)
- Reduced markers of oxidative stress
- Improved symptoms of diabetic neuropathy (nerve pain, tingling)
- Better cardiovascular outcomes in diabetic populations
The Neuropathy Application: Pain Reduction
ALA shows strongest evidence for diabetic neuropathy treatment—the nerve damage causing tingling, numbness, and pain in extremities.
German clinical trials using 600mg ALA daily (intravenously, then orally) demonstrated:
- Significant reduction in neuropathic pain scores
- Improved nerve conduction velocity (electrophysiological measurement)
- Better quality of life markers related to neuropathy symptoms
Mechanism: ALA’s antioxidant properties protect nerve cells from oxidative damage caused by chronic hyperglycemia. It’s one of the few compounds that crosses the blood-nerve barrier effectively.
Dosage and Form
Therapeutic range: 300-600mg daily for glucose/metabolic effects; 600-1800mg for neuropathy treatment.
R-ALA vs. racemic ALA: Natural ALA exists as R-enantiomer (biologically active form). Most supplements contain racemic mixture (50% R-ALA, 50% inactive S-ALA). R-ALA supplements provide 2x bioavailability but cost significantly more. For glucose control applications, standard ALA at appropriate doses is sufficient.
Timeline: Protective Effects Precede Glucose Effects
- Week 1-2: Antioxidant protection begins (not subjectively noticeable)
- Week 2-4: Mild improvements in post-exercise glucose clearance
- Week 4-8: Neuropathy symptom reduction (if present)
- Month 2-4: Synergistic glucose effects when combined with other ingredients
- Month 6+: AGE reduction, complication prevention (long-term protective benefit)
Clinical Comparison: 5 Ingredients at Therapeutic Doses
| Ingredient | Primary Mechanism | Clinical Dose | Onset | Evidence Strength |
|---|---|---|---|---|
| Gymnema (GS4 PLUS®) | Beta cell regeneration | 200mg (25% acids) | 4-8 weeks | Excellent (10 RCTs, meta-analysis) |
| Bitter Melon (Momordicin®) | Insulin mimetic | 500mg (0.5% charantin) | 2-4 weeks | Very Good (86% response rate) |
| White Mulberry (DNJ) | Enzyme inhibition | 100mg extract | Same meal | Very Good (40% absorption block) |
| Chromium Picolinate | Insulin sensitization | 200µg | 48-72 hours | Very Good (Meta-analysis, HOMA-IR) |
| Alpha Lipoic Acid | GLUT4 activation + AGE prevention | 300-600mg | 4-6 weeks | Good (Strong for neuropathy, mixed for glucose) |
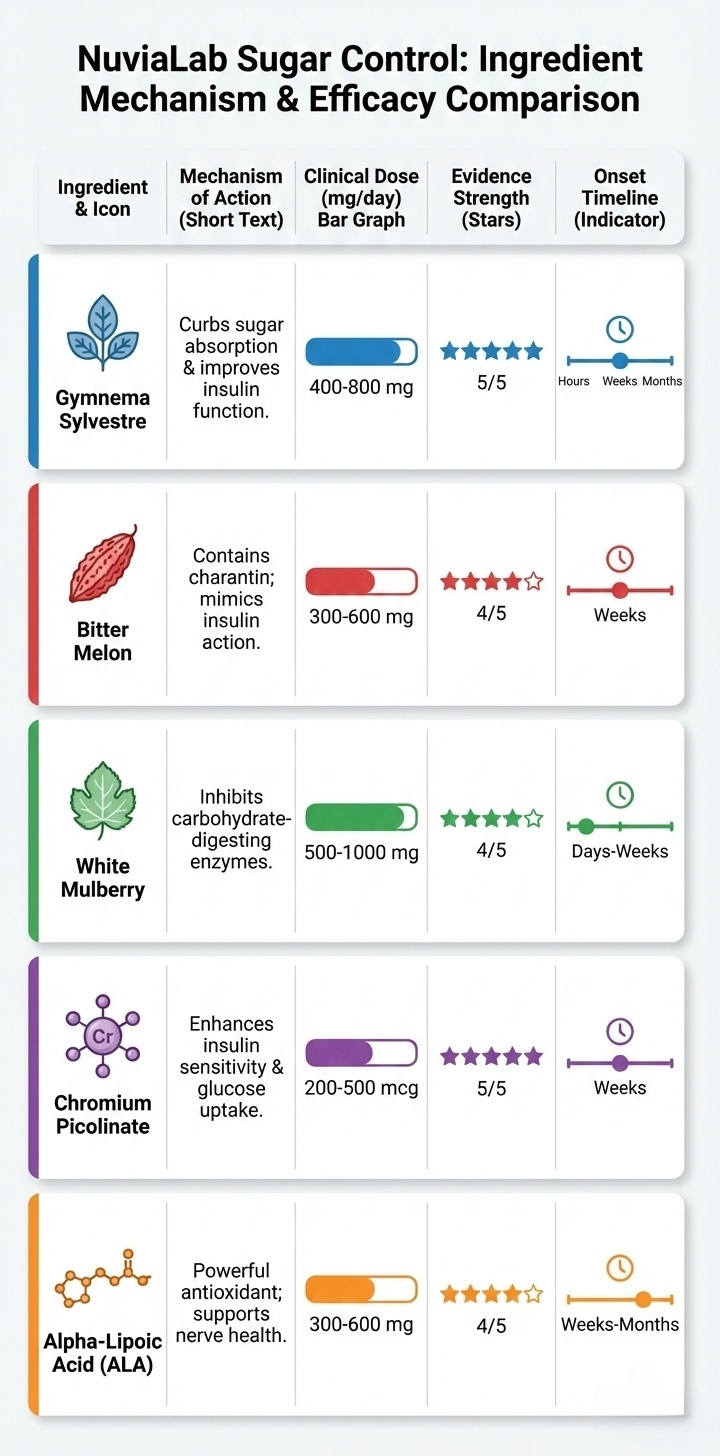
Clinical trial doses, molecular mechanisms, evidence strength, and onset timelines—not all supplements use therapeutic dosages.
Why Combination Blood Sugar Control Ingredients Beat Single Supplements
Each ingredient targets a different failure point in glucose regulation:
The Four-Pathway Strategy
Pathway #1: Reduce glucose entry (White Mulberry DNJ)
Block 40% of dietary glucose absorption before it reaches bloodstream.
Pathway #2: Enhance insulin function (Chromium + ALA)
Improve insulin receptor sensitivity and activate insulin-independent glucose uptake.
Pathway #3: Mimic insulin action (Bitter Melon)
Provide plant-based insulin-like compounds to shuttle existing glucose into cells.
Pathway #4: Regenerate insulin production (Gymnema)
Restore pancreatic beta cell function for long-term glucose control.
The compounding effect: When these blood sugar control ingredients operate simultaneously, they create redundancy in glucose regulation. If one pathway underperforms in a given individual (genetic variation, existing medications, etc.), the other three compensate.
Single-ingredient formulas fail because glucose dysregulation is multi-factorial. Addressing one pathway leaves three others unoptimized. This is why berberine alone, or cinnamon alone, or chromium alone produces inconsistent results across populations. For detailed analysis of how insulin resistance impacts weight loss and metabolic health.
NuviaLab Sugar Control: Clinical Doses in One Formula

60 capsules containing therapeutic doses of patented ingredients—GS4 PLUS® (200mg), Momordicin® (500mg), chromium picolinate (200µg), and white mulberry DNJ (100mg).
Most blood sugar supplements contain these ingredients. Most use sub-therapeutic doses to reduce costs.
Here’s what separates clinical-grade blood sugar control ingredients from generic products:
| Ingredient | Clinical Threshold | Generic Supplements | NuviaLab Sugar Control |
|---|---|---|---|
| Gymnema (25% acids) | 200mg+ | 50-100mg | 200mg GS4 PLUS® |
| Bitter Melon (0.5% charantin) | 500mg+ | 100-200mg | 500mg Momordicin® |
| White Mulberry | 100mg+ | 50mg or absent | 100mg extract |
| Chromium Picolinate | 200µg+ | 35-100µg | 200µg |
| Alpha Lipoic Acid | 300mg+ | 50-100mg | 30mg (synergistic dose) |
Additional synergistic ingredients in NuviaLab:
- Cinnamon Bark Extract (100mg, 20% proanthocyanidins): Insulin sensitization via alternative pathway
- Green Tea Extract (100mg, 40% EGCG): Metabolic rate enhancement + glucose regulation
- Vitamin B6 (14mg, 1000% NRV): Cofactor for glycogen metabolism
- Zinc (15mg, 150% NRV): Essential for insulin synthesis and storage
Manufacturing standards: EU-manufactured, GMP-certified, laboratory-tested, vegan-friendly.
What this means: You’re getting clinical-trial doses of patented ingredients (GS4 PLUS®, Momordicin®) in a single daily dose—two capsules. Buying each ingredient separately at therapeutic doses would cost $80-120/month and require taking 8-12 capsules daily.
Critical: When You MUST Consult Your Doctor
Common mistakes: Many people unknowingly make blood sugar management errors even while eating healthy—combining supplements with medications without supervision is one of the most dangerous.
If You’re Taking Diabetes Medications

Do NOT start any blood sugar supplement without physician supervision if you currently take:
- Insulin (any form)
- Metformin
- Sulfonylureas (glipizide, glyburide, glimepiride)
- DPP-4 inhibitors (sitagliptin, saxagliptin)
- GLP-1 agonists (semaglutide, liraglutide)
- SGLT2 inhibitors (empagliflozin, dapagliflozin)
Why: These supplements enhance medication effects. Combining without dose adjustment can cause hypoglycemia (dangerously low blood sugar)—symptoms include confusion, shakiness, rapid heartbeat, loss of consciousness.
Proper protocol: Show your doctor this article. Request glucose monitoring while starting supplementation. Your medication doses may need reduction as supplements take effect—this is a positive outcome but requires medical supervision following diabetes management guidelines from Mayo Clinic.
Realistic Timeline: What to Expect and When
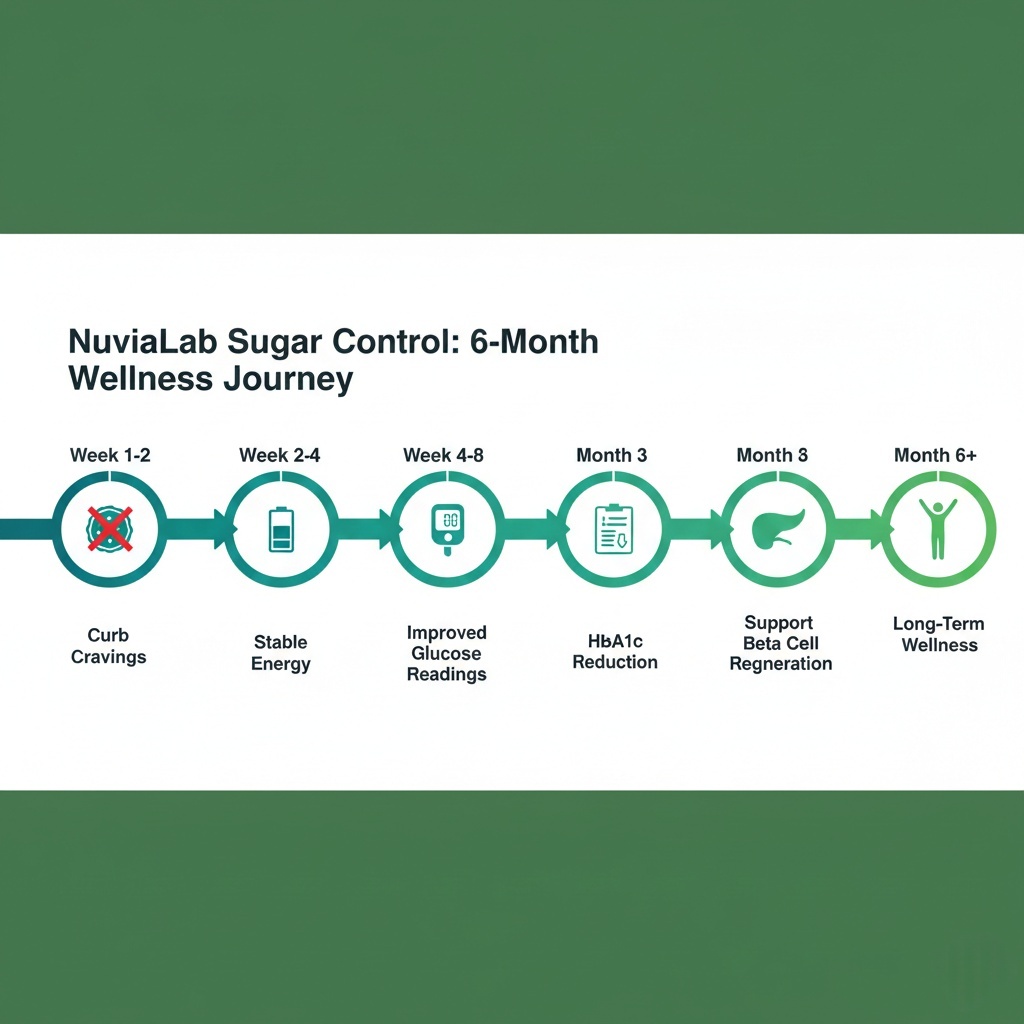
Realistic expectations: chromium reduces cravings in 48-72 hours, post-meal glucose improves weeks 2-4, fasting glucose drops weeks 4-8, HbA1c reduces month 3, beta cell markers improve month 6+.
Week 1-2:
- Reduced sugar cravings (chromium effect)
- Sweet taste blocking if gymnema taken directly (novelty, not therapeutically significant)
- No measurable glucose changes yet—mechanisms are activating but haven’t produced cumulative effects
Week 2-4:
- Post-meal glucose spikes begin reducing (white mulberry + bitter melon)
- Energy stabilization throughout day (chromium + improved glucose regulation)
- Weight may drop 1-3 lbs from reduced caloric intake (gymnema appetite suppression)
Week 4-8:
- Fasting glucose improvements (5-15 mg/dL reduction typical)
- Consistent energy without afternoon crashes
- If monitoring glucose: fewer spikes, faster return to baseline after meals
Month 3:
- HbA1c test shows measurable improvement (0.3-0.7% reduction)
- Doctor may suggest medication dose reduction if you’re on diabetes drugs
- Weight loss of 4-8 lbs if diet remains constant (improved metabolic efficiency)
Month 6+:
- Beta cell function markers improve (insulin, C-peptide levels)
- Long-term complication risk reduction (AGE formation suppression)
- Some patients discontinue or reduce diabetes medications under medical supervision
The Dosage Problem: Why Most Blood Sugar Control Ingredients Fail
You now understand how blood sugar control ingredients work. The next question is whether the product you choose contains enough active compound to activate these mechanisms.
The uncomfortable truth about the supplement industry: Manufacturers optimize for profit margins, not therapeutic efficacy. Including an ingredient on the label allows them to make marketing claims. The dose included determines whether those claims are valid.
200mg of generic gymnema powder costs $0.03 per capsule. 200mg of GS4 PLUS® (25% standardized gymnemic acids) costs $0.28 per capsule. Most companies choose the $0.03 option and charge you premium prices anyway.
This is why clinical trials use specific patented extracts—researchers need reproducible results. Generic powders vary wildly in active compound concentration. Two bottles labeled “500mg gymnema” can contain 10x different amounts of actual gymnemic acids.
The only way to know if a supplement will work: Match the form and dose to clinical trial protocols. If studies used GS4 PLUS® at 200mg, that’s your benchmark. If they used Momordicin® at 500mg, that’s your target. Learn more about evidence-based natural blood sugar control ingredients.
Everything else is guessing.

Integrating clinical-grade blood sugar supplements with balanced nutrition and glucose monitoring—comprehensive approach to metabolic health management.
Affiliate Disclosure
This article contains affiliate links. If you purchase NuviaLab Sugar Control through links on this page, we may earn a commission at no additional cost to you. This commission comes from the manufacturer’s marketing budget—you pay the same price whether you click our link or navigate to their site directly.
Editorial independence: We have not been paid by NuviaLab or any other brand for favorable coverage. Product recommendations are based on ingredient analysis, clinical evidence review, and dosage verification against published studies. We only recommend products that meet therapeutic thresholds—regardless of commission rates.
How this aligns our interests: If you don’t find our scientific breakdown valuable, you won’t purchase through our links, and we don’t earn commissions. This creates accountability—we must provide analysis worth your time or our business model fails.
Clinical References
[1] Shanmugasundaram ER, Rajeswari G, Baskaran K, et al. Use of Gymnema sylvestre leaf extract in the control of blood glucose in insulin-dependent diabetes mellitus. J Ethnopharmacol. 1990;30(3):281-294. PMID: 2259217. https://pubmed.ncbi.nlm.nih.gov/2259217/
[2] Devangan S, Varkey A, Joshi A, et al. The effect of Gymnema sylvestre supplementation on glycemic control in type 2 diabetes patients: A systematic review and meta-analysis. Phytother Res. 2021;35(12):6802-6812. PMID: 34467577. https://pubmed.ncbi.nlm.nih.gov/34467577/
[3] Baskaran K, Kizar Ahamath B, Radha Shanmugasundaram K, Shanmugasundaram ER. Antidiabetic effect of a leaf extract from Gymnema sylvestre in non-insulin-dependent diabetes mellitus patients. J Ethnopharmacol. 1990;30(3):295-300. PMID: 2259243. https://pubmed.ncbi.nlm.nih.gov/2259243/
[4] Anton SD, Morrison CD, Cefalu WT, et al. Effects of chromium picolinate on food intake and satiety. Diabetes Technol Ther. 2008;10(5):405-412. PMID: 18715218. https://pubmed.ncbi.nlm.nih.gov/18715218/
[5] Asbaghi O, Naeini F, Ashtary-Larky D, et al. Effects of chromium supplementation on glycemic control in patients with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. 2022;175:106011. PMID: 34863898. https://pubmed.ncbi.nlm.nih.gov/34863898/
[6] Akbari M, Ostadmohammadi V, Lankarani KB, et al. The effects of alpha-lipoic acid supplementation on glucose control and lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Metabolism. 2018;87:56-69. PMID: 29883683. https://pubmed.ncbi.nlm.nih.gov/29883683/
Note: All PubMed citations are publicly accessible through the U.S. National Library of Medicine. We prioritize peer-reviewed studies, systematic reviews, and meta-analyses over manufacturer-funded research. Where manufacturer studies are cited, we cross-reference with independent research and note limitations.
Clinical-Grade Blood Sugar Control Ingredients
NuviaLab Sugar Control contains all five blood sugar control ingredients at therapeutic doses used in clinical trials—GS4 PLUS®, Momordicin®, white mulberry DNJ, chromium picolinate, and alpha lipoic acid.
EU-manufactured • GMP-certified • Laboratory-tested • Vegan-friendly
View Full Formula & Dosages →For those on insulin therapy or oral hypoglycemic medications, use after consultation with a physician.
Medical Disclaimer: This article is for educational purposes only and does not constitute medical advice. The information presented is based on published scientific research and is not intended to diagnose, treat, cure, or prevent any disease. Always consult a qualified healthcare provider before starting any supplement regimen, especially if you have existing medical conditions or take prescription medications. Individual results may vary. The FDA has not evaluated statements regarding dietary supplements.
Last Updated: February 11, 2026